 
However, despite a number of modifications to the model, by the early 1920s Bohr’s model seemed to be a dead end, as efforts to generalize the model. Atomic models have gone through many changes over time, evolving as necessary to fit experimental data. 
The orbit with n = 1 is the lowest lying and most tightly bound. Atomic model, in physics, a model used to describe the structure and makeup of an atom. He concluded that electron will have more energy if it is located away from the nucleus whereas electrons will have less energy if it located near the nucleus. All features of Bohr’s model of the atom can be summarized in Bohr’s Postulates. An atom is made of tiny negatively charged. In atomic physics, the Bohr model of the atom (also known as the Rutherford-Bohr model) is the modern model of the hydrogen atom introduced by Danish physicist Niels Bohr. Atoms can be broken down into smaller parts. 
In his final years, he devoted himself to the peaceful application of atomic physics and to resolving political problems arising from the development of atomic weapons.Īs n decreases, the energy holding the electron and the nucleus together becomes increasingly negative, the radius of the orbit shrinks and more energy is needed to ionize the atom. According to the Bohr Atomic model, a small positively charged nucleus is surrounded by revolving negatively charged electrons in fixed orbits. Year: Scientist(s) New evidence: Changes to the atomic model: 1897: Thomson: The discovery of electrons. (b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project. In fact Nield found that the ratio of energy in electrons and the frequency of their orbits around the nucleus was equal to Planck's constant (the proportion of light's energy to its wave frequency, or approximately 6.626 x 10-23 ).\): The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Learn the basic features of Bohr’s model of the atom, such as the nucleus, the electrons, the orbits and the energy levels. Bohr’s model of the hydrogen atom started from the planetary model, but he added one assumption regarding the electrons. This approach allowed Niels to explain a relatively stable atomic Model, which still hinged on the orbital model that Rutherford postulated. The first part of his theory states that all matter is made of atoms, which are indivisible. Dalton based his theory on the law of conservation of mass and the law of constant composition. Niels Bohr which after his PhD circa 1912 had joined Rutherford realized that Rutherford's model wasn't quite right and started to articulate a model that borrowed from Planck's quantum theory. Daltons atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties. All of the negative charge was held in the electrons, orbiting the dense nucleus like planets around the sun. This model depicted an atomic model with nearly all its mass, and positive charge, in a central nucleus about 10,000 times smaller than the atom itself. Rutherford experiment with alpha particles shot at a thin gold foil resulted in the Rutherford model of the atom (Orbital Model). The Atom: + 1 amu nucleus Proton outside nucleus - 1/2000 amu Electron Neutron none 1 amu nucleus. At the end of the learning period, student should be able to: a. Bohr Rutherford Atomic Model: Bohr & Rutherford combined their theories to create the current simplified atomic model The atom is made up of three sub-atomic particles: Protons Electrons Neutrons. Thomson actually) which blew a big hole in "Plum Pudding Hypothesis" of the atom. How did Neils Bohr explain the stability of atom (b) An element has an atomic number of 11 and its mass number is 23. Describe how the Bohr Model of the atom improved Rutherford’s Atomic Model. Thomson that resulted "Plum Pudding" model of the atom and the 2nd one by Rutherford (a student of J.J. He concluded that electron will have more energy if it is located away from the nucleus whereas the electrons will have less energy if it located near the nucleus. What was thought of as a single particle about 1 ×. 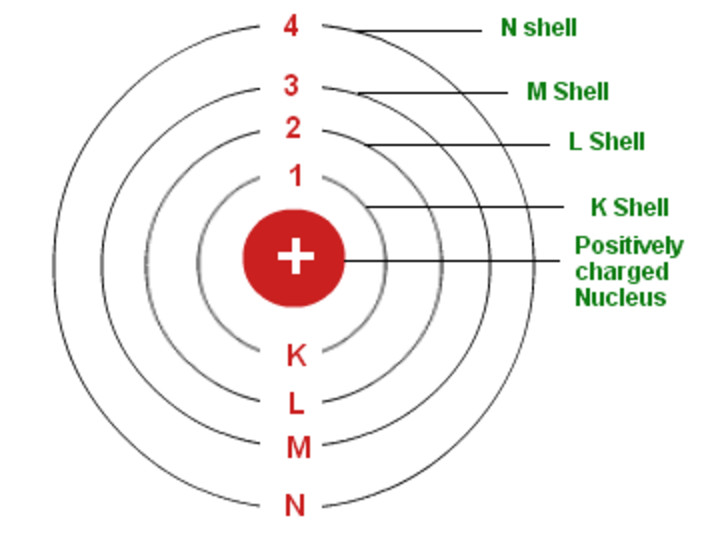
The idea of the atom as the building block of matter has developed over time. (Image credit: Pixabay) The Bohr model, introduced by Danish physicist Niels Bohr in. A model of an atom according to Niels Bohr. Well there were two experiments back to back one by J.J. According to Bohr Atomic model, a small positively charged nucleus is surrounded by revolving negatively charged electrons in fixed orbits. GCSE AQA Trilogy Models of the atom - AQA Developing the atom. The Bohr model is neat, but imperfect, depiction of atom structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
